+273% 1-year stock performance, January 7, 2013 to January 7, 2014, no insider sales
+135% 1-month (roughly) performance, December 9,
2013 to January 7, 2014, no insider sales
-3% 1-week performance, December 31, 2013 to January 7, 2014, no insider sales
There are no sale-related Form 4 filings on the
SEC website for Provectus filings through Tuesday, January 7th. For the previously provided link, check the “include” circle under “Ownership?” to bring up stock
ownership-related filings. Another way is here, where I searched for filings using "4." I note Tuesday because filings must
be made within two business days. It is theoretically possible management sold shares Wednesday or Thursday, with reporting of these transactions made via filings Friday and Monday, respectively. While I cannot write with certainty
management have not sold shares, I’m going to go out on a limb and suggest they have not.
Historically, there have been no insider sales by Provectus officers and directors.* There only have been purchases.
You may recall Provectus' stock’s value proposition
from my
September 22nd investment letter (the closing share price: “The Company’s stock value proposition is very compelling at the
current share price. Provectus appears close to achieving regulatory clarity
for its lead indication, appears close to consummating regional license
transactions for PV-10 in China and India, appears to have as least the
interest of Pfizer as well as the attention of other global pharmaceutical
companies, has no historical insider
selling and plenty of historical insider buying, and has low
institutional ownership. The Company, on the other hand, has a stock that until
this year was mired in a multi-year downward trend, a capital structure that
historically has weighed on the share price, trades on a minor U.S. stock
exchange, is led by first-time public company leadership with no prior
experience bringing an oncology drug to market and of which Wall Street is very
skeptical, and has no institutional or Wall Street sponsor (the lack of playing
Wall Street’s game in a highly regulated and capital-intensive industry like
biotechnology, together with some shortcomings, has restrained the share price
heretofore).”
Buy/sell transactions by directors/ex-directors
(chronologically, I believe, Fuchs, McMasters, Koe and Smith) provide
information and sometimes knowledge. I believe I am correct in writing only Koe purchased stock
as a director (part of the September announced private placements), already having considerable ownership prior to
his election as a member of Provectus' board of directors. Fuchs’ ownership, I believe,
derived from his early funding involvement with the company, with warrants
and possibly stock that also may have come with it. All directors have been awarded stock
options over time for their roles.
* With Fuchs' departure from the board in July 2011, thus becoming an ex-director, he may have sold some or all of his shares thereafter.
* With Fuchs' departure from the board in July 2011, thus becoming an ex-director, he may have sold some or all of his shares thereafter.
 |
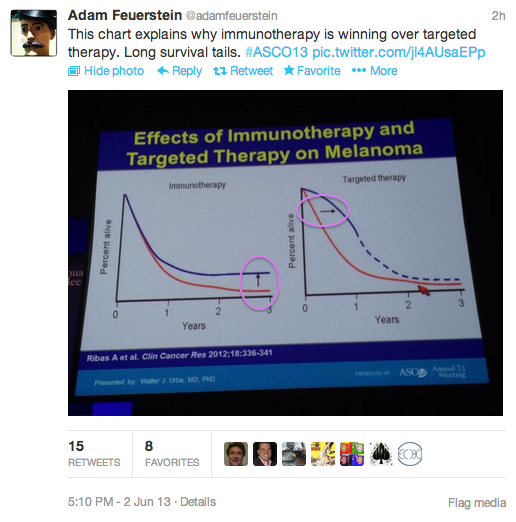
| A tweet from Thursday |
Management has told us they await FDA guidance, per their December 18th Provectus Type C Meeting With FDAOncology Division Held December 16, 2013 press release. Presume management knew what went on in the meeting by being and participating in the
meeting. Then:
- If the meeting went “bad,” would they not have sold shares by now? By not selling, are they, along with shareholders, planning to go down with the ship and stock? That's, um, noble...
- If the meeting (and, potentially, any follow-up**), however, was good or great, would it not make sense to continue to own stock, given now their view into FDA guidance that also might inform their view of PV-10's commercialization timeline. The founding principals and principal no. 4 are playing for tens and tens and tens and … of dollars, having historically eschewed a Wall Street or investment sponsor, and desirous of leading and controlling the company and the solving of a problem of global importance. Management is not playing for tens and tens of cents.
** It's clear from the thousands of pages of administrative and correspondence documents readily available on the Internet for drug approvals that sponsors (applicants) and the FDA routinely and regularly communicate and interact through the drug approval process.
From my investment letter: "PV-10, a novel oncology compound being developed by Knoxville, Tennessee-based Provectus Biopharmaceuticals, Inc., exemplifies innovation over incrementalism, meaningful over marginal, productized technology over hypothetical, and changing the world over accepting the status quo, with not an insignificant amount of serendipity over contrivance. In sum, these form the quintessential essence of a paradigm shift in the treatment of cancer.
From my investment letter: "PV-10, a novel oncology compound being developed by Knoxville, Tennessee-based Provectus Biopharmaceuticals, Inc., exemplifies innovation over incrementalism, meaningful over marginal, productized technology over hypothetical, and changing the world over accepting the status quo, with not an insignificant amount of serendipity over contrivance. In sum, these form the quintessential essence of a paradigm shift in the treatment of cancer.
This is where my investment thesis begins and ends: a novel
drug compound with a pristine safety profile, a treatment well tolerated by and
easily administered to patients, a ready made product inexpensively produced at
scale, and a vast addressable market of unmet need that should be fully and
very profitably met over time." Bold emphasis is mine. No one can call PV-10 a cure (or the Holy Grail), for now. I focus on the drug's ability, it's vast potential, to shift how
doctors think about and subsequently act on treating cancer once found.
Cancer causes disfigurement through surgery, often the first
solution considered by physicians (for those patients where surgery is a viable
option) for many solid tumor cancers once discovered and diagnosed. PV-10’s
tissue sparing ability, together with its safety and efficacy, eventually should eliminate the consideration of surgery first.
Cancer causes discomfort of varying degrees, typically adverse
to toxic, when chemotherapy, radiotherapy, and many immunotherapies with actual or
likely black box warnings or detrimental short- and long-term effects are used. PV-10’s 30-minute “half-life” means the drug
is in the patient’s bloodstream for 30 minutes before it is excreted in the
bile. Much more effective than other therapies, PV-10’s
pristine safety profile and paucity of side effects eventually should eliminate the need
for considering chemotherapy, radiotherapy or immunotherapy before first using PV-10.
Cancer causes death, often because the patient’s tumor
burden is too great for the immune system to effectively fight back. Cancer
wins because it’s discovered too late. It wins because the immune system is
overwhelmed by toxic therapies like chemotherapy, radiotherapy and many
immunotherapies, rendering it unable to effectively fight back. Can PV-10 mitigate or eliminate death caused by cancer? Time, more study and
research, etc. only will tell. In the case of heavily diseased or tumor burdened patients, PV-10 eventually might be injected into as many
accessible tumors as possible with the drug, increasing the likelihood of as
many antigens as possible being presented to T cells and other immune system
components for the immune system to eradicate as many or all vestiges of cancer
from the body. For those previously with no hope, patients with very heavily diseased burden and
severely compromised immune systems, PV-10 still could be used in combination (to be further examined, strategized, optimized and ultimately decided) with other
systemic therapies like chemotherapy, radiotherapy and other immunotherapies to reduce tumor burden sufficiently for PV-10 to then help stimulate the
immune system to finish off the cancer.
A paradigm shift in the treatment of cancer
Indeed.